Embryos kill off male tissue to become female
Add a new ingredient to the sugar, spice and everything nice needed to make girls.
A protein called COUP-TFII is necessary to eliminate male reproductive tissue from female mouse embryos, researchers report in the Aug. 18 Science. For decades, females have been considered the “default” sex in mammals. The new research overturns that idea, showing that making female reproductive organs is an active process that involves dismantling a primitive male tissue called the Wolffian duct.
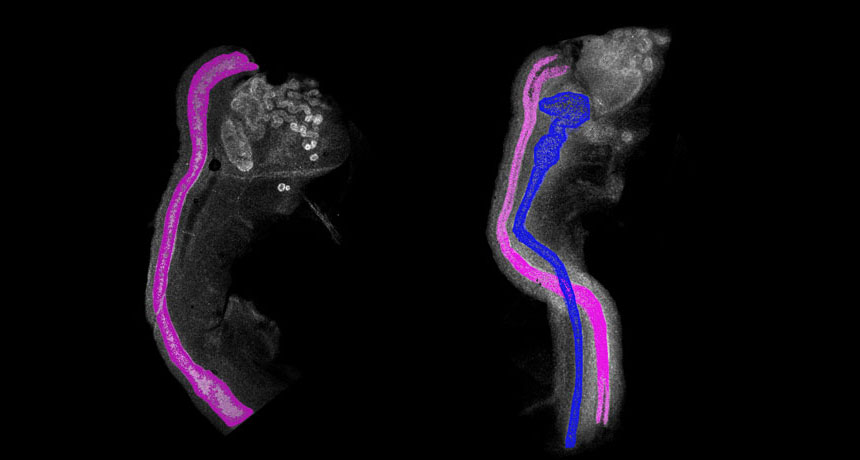
In males, the Wolffian duct develops into the parts needed to ejaculate sperm, including the epididymis, vas deferens and seminal vesicles. In females, a similar embryonic tissue called the Müllerian duct develops into the fallopian tubes, uterus and vagina. Both duct tissues are present in early embryos.
A study by French endocrinologist Alfred Jost 70 years ago indicated that the testes make testosterone and an anti-Müllerian hormone to maintain the Wolffian duct and suppress female tissue development. If those hormones are missing, the Wolffian duct degrades and an embryo by default develops as female, Jost proposed.
That’s the story written in textbooks, says Amanda Swain, a developmental biologist at the Institute of Cancer Research in London. But the new study “demonstrates that females also have a pathway to make sure you don’t get the wrong ducts,” says Swain, who wrote a commentary in the same issue of Science.
Testing Jost’s hypothesis wasn’t what reproductive and developmental biologist Humphrey Yao and colleagues set out to do. Instead, the researchers wanted to learn how tissues on the outside of the early ducts communicate with the tubes’ lining, says Yao, of the National Institute of Environmental Health Sciences in Research Triangle Park, N.C.
The COUP-TFII protein is produced in that outer layer, and Yao suspected it was involved in talking with the lining. The researchers blocked the communication in early female mouse embryos’ reproductive tissue by removing the gene that produces COUP-TFII.
To the team’s surprise, the Wolffian duct remained in the female mice along with the female Müllerian duct. That shouldn’t happen, according to the textbooks. “We were just scratching our heads,” Yao says.
Searching for an explanation, Yao and colleagues first tested whether removing COUP-TFII changed the ovaries to produce testosterone like testes do. Testosterone could feed the male tissue and allow it to persist, the researchers thought.
“No, the ovary is just like an ovary. There’s nothing wrong with it,” Yao says. “We were just shocked. This can’t be happening.” Further experiments demonstrated that no stray testosterone was responsible for the male tissue sticking around.
Instead, COUP-TFII appears to be the foreman of a biochemical wrecking crew that demolishes the Wolffian duct in females. Without the protein barking orders, the demolition crew is idle and the male duct isn’t torn down. Signals that trigger COUP-TFII production and activity aren’t yet understood.
“This study fills a void in our understanding of the mechanism of regression of the Wolffian duct,” reproductive biologists Patricia Donahoe and David Pepin of Harvard Medical School said in an e-mail. More research is needed to understand how the protein interacts with male hormones to regulate reproductive tract development, they say.
While the study used mice, COUP-TFII probably works the same way in other mammals, including humans, Donahoe says. Females rarely still carry Wolffian duct remnants, sometimes leading to tumors. The opposite sometimes happens, too, resulting in males with female reproductive organs. Those men may be infertile and have other problems, such as cysts. Researchers should look for defects in COUP-TFII in patients with reproductive problems, Donahoe says.